NESTcc Research Network
We collaborate with leading health organizations to curate high-quality, real-world data from multiple sources across 220 million patient records.
Joining the NEST Marketplace provides an unparalleled opportunity for any partners involved in RWE support for the medical device industry.





The NEST community is passionately committed to transforming the way medical device technologies are tested, approved and monitored.
We envision a world in which people are empowered to make informed medical choices that enable patients to live their lives to the fullest extent possible.
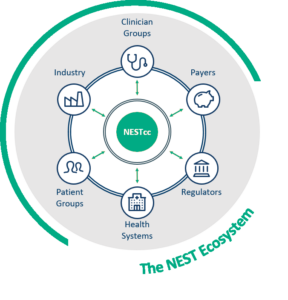
We collaborate with leading health organizations to curate high-quality, real-world data from multiple sources across 220 million patient records.
See NESTcc’s current open calls for opportunities to collaborate or provide input on our initiatives.
Learn more about our capabilities and consult with our team on using RWE to advance your organization’s objectives.
National Evaluation System for health Technology Coordinating Center (NESTcc)
1655 N Ft. Myer Dr, 12th Floor, Arlington, VA 22209
© Copyright 2024 | Privacy Policy
This site uses cookies to improve your browsing experience; the website may not work as expected without them. By continuing to browse this site, you consent to the use of cookies.
OKLearn moreWe may request cookies to be set on your device. We use cookies to let us know when you visit our websites, how you interact with us, to enrich your user experience, and to customize your relationship with our website.
Click on the different category headings to find out more. You can also change some of your preferences. Note that blocking some types of cookies may impact your experience on our websites and the services we are able to offer.
These cookies are strictly necessary to provide you with services available through our website and to use some of its features.
Because these cookies are strictly necessary to deliver the website, refusing them will have impact how our site functions. You always can block or delete cookies by changing your browser settings and force blocking all cookies on this website. But this will always prompt you to accept/refuse cookies when revisiting our site.
We fully respect if you want to refuse cookies but to avoid asking you again and again kindly allow us to store a cookie for that. You are free to opt out any time or opt in for other cookies to get a better experience. If you refuse cookies we will remove all set cookies in our domain.
We provide you with a list of stored cookies on your computer in our domain so you can check what we stored. Due to security reasons we are not able to show or modify cookies from other domains. You can check these in your browser security settings.
We also use different external services like Google Webfonts, Google Maps, and external Video providers. Since these providers may collect personal data like your IP address we allow you to block them here. Please be aware that this might heavily reduce the functionality and appearance of our site. Changes will take effect once you reload the page.
Google Webfont Settings:
Google Map Settings:
Google reCaptcha Settings:
Vimeo and Youtube video embeds:

NESTcc posts Request for Participation 23-MP1001
/in NewsRequest for Participation 23-MP1001 Expanding on previous work, the objective of this Request for Participation is to establish additional strategic partners to form the NEST Marketplace. The Marketplace and its partners will be able to efficiently curate, aggregate, and analyze health data, and will contribute to advancing the capabilities of the NEST Marketplace. This Request for […]
NESTcc posts Request for Proposal (RFP) 23-AS1001
/in NewsNational Evaluation System for health Technology Coordinating Center (NESTcc) Releases a Request for Proposal Relating to its Medical Device Active Surveillance Program NESTcc’s Active Surveillance Program for Medical Devices takes the next step ARLINGTON, Va.–(BUSINESS WIRE)–The National Evaluation System for health Technology Coordinating Center (NESTcc), an initiative of the Medical Device Innovation Consortium (MDIC), announced […]
NESTcc posts Request for Information (RFI) 23-001
/in NewsNational Evaluation System for health Technology Coordinating Center posts Request for Information (RFI) 23-001 for Medical Device Real-World Evidence Marketplace June 08, 2023 ARLINGTON, Va.–(BUSINESS WIRE)–The National Evaluation System for health Technology Coordinating Center (NESTcc), an initiative of the Medical Device Innovation Consortium (MDIC), is soliciting the interest of organizations to partner in generating high-quality […]