Case Study: Digital Twinning & Additive Manufacturing in Microfluidics at BioSens8
Advanced Manufacturing in Action Case Study
Advanced manufacturing offers an alternative to traditional labor-intensive processes—helping reduce cost, improve quality, contribute to sustainability efforts, and shorten development time.
This case study from MDIC’s Advanced Manufacturing Clearing House explores how digital twinning and additive manufacturing were applied to the design and optimization of a microfluidic system for biosensors.
BioSens8's Digital Transformation At-a-Glance
Microfluidic device development requires precise control of fluid behavior at the micron scale—often leading to high fabrication costs, long iteration cycles, and limited visibility into internal system performance.
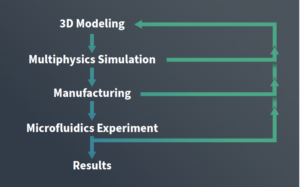
By combining simulation-based digital twins with additive manufacturing, teams can evaluate designs in a physics-based environment before fabrication—reducing reliance on trial-and-error prototyping and enabling faster, more practical iteration.
This case study outlines a simulation-aided approach to microfluidics design and manufacturing, including:
- Application of digital twinning to model fluid flow, heat transfer, and mass transport
- Use of additive manufacturing to rapidly produce and test prototypes
- An iterative workflow combining simulation, fabrication, and experimental validation
- Quantitative performance metrics used to evaluate and optimize design
The combined use of digital twinning and additive manufacturing enabled:
- Reduced time between design iterations—from months to weeks
- Decreased cost and material waste during development
- Improved understanding of internal system behavior through simulation
- A more efficient pathway for developing and testing microfluidic devices