Project Goal:
Health care networks with well developed, integrated data systems are able to generate real-world data accepted by the FDA for regulatory submissions.
Health care networks with well developed, integrated data systems are able to generate real-world data accepted by the FDA for regulatory submissions.
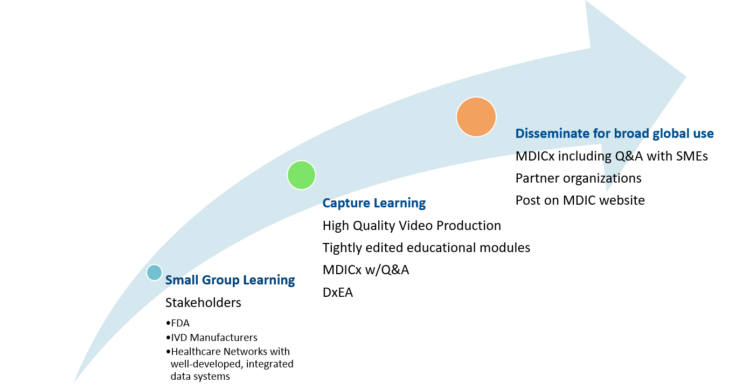
MDIC recently released its Real-World Clinical Evidence Generation: Advancing Regulatory Science and Patient Access for In Vitro Diagnostics (IVDs) framework created collaboratively with representatives from FDA and IVD manufacturers. Another project, Systemic Harmonization and Interoperability Enhancement for Laboratory Data (SHIELD) has been working for years laying out the foundation for linking IVD test data to the laboratory information systems and then into the institutional EHR. The current coronavirus pandemic has many diagnostic tests in the marketplace that have Emergency Use Authorization (EUA) status which allows them on the market without a full FDA submission. This project will bring the IVD manufacturers, FDA, and hospitals together to use IVD Real-World Evidence (RWE) in regulatory systems.

 EFS Express Archive
EFS Express Archive